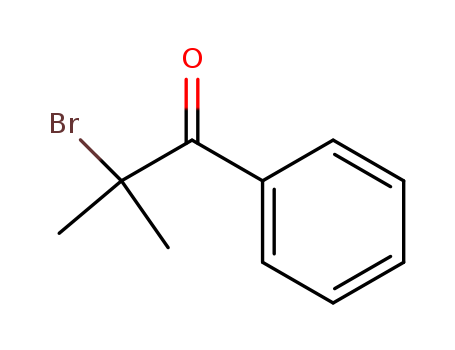
10409-54-8
- Product Name:2-BROMO-2-METHYLPROPIOPHENONE
- Molecular Formula:C10H11BrO
- Purity:99%
- Molecular Weight:227.101
Product Details;
CasNo: 10409-54-8
Molecular Formula: C10H11BrO
Appearance: clear yellow liquid
Buy Quality 2-BROMO-2-METHYLPROPIOPHENONE 10409-54-8 with Efficient Delivery
- Molecular Formula:C10H11BrO
- Molecular Weight:227.101
- Appearance/Colour:clear yellow liquid
- Vapor Pressure:0.0188mmHg at 25°C
- Refractive Index:n20/D 1.5561(lit.)
- Boiling Point:253 °C at 760mmHg
- Flash Point:48.2 °C
- PSA:17.07000
- Density:1.358 g/cm3
- LogP:3.04280
2-BROMO-2-METHYLPROPIOPHENONE(Cas 10409-54-8) Usage
|
Chemical Properties |
clear yellow liquid |
| Description | 2-Bromo-2-methylpropiophenone plays a crucial role as a chemical intermediate, offering versatility in organic synthesis and finding essential applications in pharmaceuticals, fine chemicals, and other industries. It is particularly employed in the production of pharmaceuticals. |
| Usage and Applications | 2-Bromo-2-methylpropiophenone is commonly used as a chemical intermediate in organic synthesis. |
| Versatility in Organic Synthesis | 2-Bromo-2-methylpropiophenone is versatile and can be utilized in the synthesis of a wide range of organic compounds due to its functional groups. |
InChI:InChI=1/C10H11BrO/c1-10(2,11)9(12)8-6-4-3-5-7-8/h3-7H,1-2H3
10409-54-8 Relevant articles
REACTION OF ALDEHYDES AND KETONES WITH t-BUTYL BROMIDE-DIMETHYL SULPHOXIDE
Armani, E.,Dossena, A.,Marchelli, R.,Casnati, G.
, p. 2035 - 2040 (1984)
Reacting aldehydes and ketones with the ...
β-Keto phosphonic esters Communication 12. Structures of the products of the reaction of some α-halo ketones of the aromatic series with triethyl phosphite and with sodium diethyl phosphite
Arbuzov B.; Vinogradova V.; Polezhaeva N.; Shamsutdinova A.
-
The reactions of 2-bromo-2-methylpropiophenone, in which the bromine is on a tertiary carbon atom, with triethyl phosphite and with sodium diethyl phosphite take the same direction with formation of diethyl 2-methyl-1-propenyl phosphate.
Enantioselective Biocatalytic Reduction of 2 H-1,4-Benzoxazines Using Imine Reductases
Zumbr?gel, Nadine,Machui, Paul,Nonnhoff, Jannis,Gr?ger, Harald
, p. 1440 - 1447 (2019)
A biocatalytic reduction of 2H-1,4-benzo...
Synthesis and Spontaneous Resolution by Crystallization of R,S-(+/-)-Dimethyl(1-methyl-2-oxo-2-phenylethyl)sulphonium Bromide. X-Ray Structure and Absolute Configuration of the R-Enantiomer
Dossena, Arnaldo,Marchelli, Rosangela,Armani, Elisabetta,Fava, Giovanna Gasparri,Belicchi, Marisa Ferrari
, p. 1196 - 1197 (1983)
R,S-(+/-)-Dimethyl(1-methyl-2-oxo-2-phen...
Direct synthesis of α-bromoketones from alkylarenes by aerobic visible light photooxidation
Tada, Norihiro,Ban, Kazunori,Hirashima, Shin-Ichi,Miura, Tsuyoshi,Itoh, Akichika
, p. 4701 - 4704 (2010)
The direct synthesis of α-bromoketones f...
10409-54-8 Process route
-
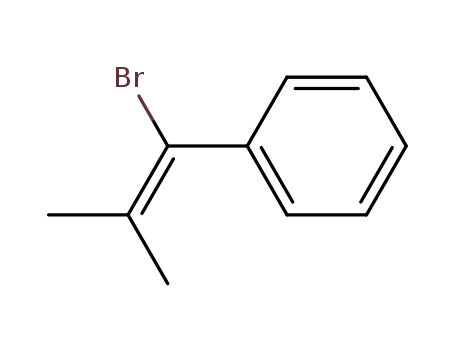
- 5912-93-6
1-bromo-2-methyl-1-phenyl-1-propene

-
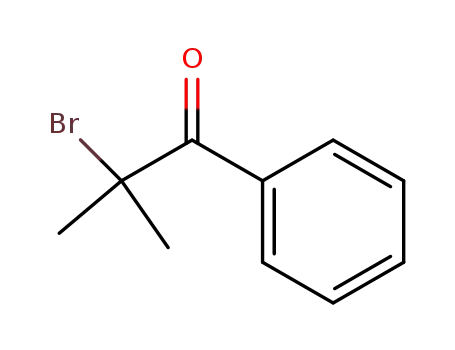
- 10409-54-8
2-bromo-2-methyl-1-phenyl-propan-1-one
| Conditions | Yield |
|---|---|
|
With [hydroxy(tosyloxy)iodo]benzene; toluene-4-sulfonic acid; In acetonitrile; at 40 ℃; for 15h; Inert atmosphere;
|
80% |
-
- 2234-23-3
C17H20O

-

- 10409-54-8
2-bromo-2-methyl-1-phenyl-propan-1-one
| Conditions | Yield |
|---|---|
|
With p-nitrobenzenesulfonamide; hydrogen bromide; oxygen; sodium nitrite; In water; acetonitrile; at 0 - 60 ℃; for 24.5h; under 760.051 Torr;
|
89% |
10409-54-8 Upstream products
-
611-70-1
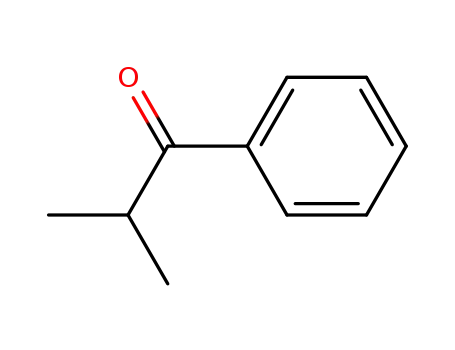
phenyl isopropyl ketone
-
20769-85-1
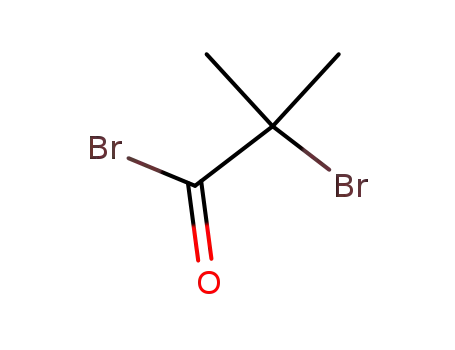
2-bromoisobutyric acid bromide
-
71-43-2
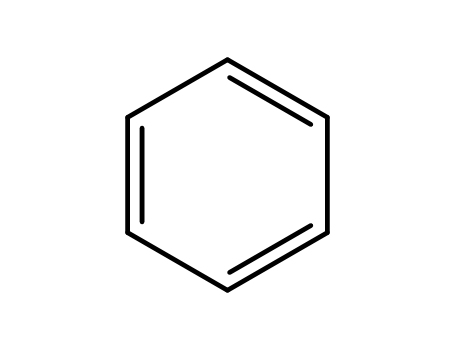
benzene
-
768-49-0
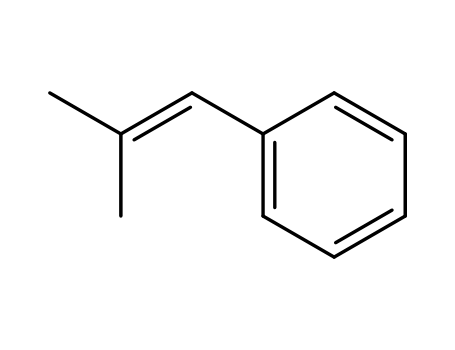
(2-methyl-1-propenyl)-benzene
10409-54-8 Downstream products
-
101284-35-9
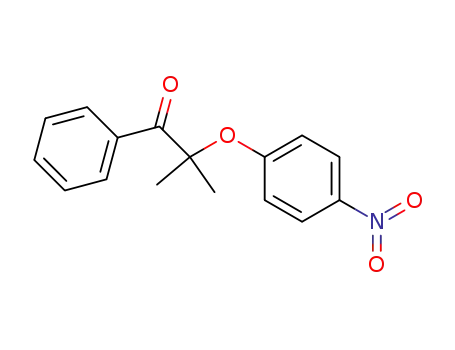
2-methyl-2-(4-nitro-phenoxy)-1-phenyl-propan-1-one
-
13605-61-3
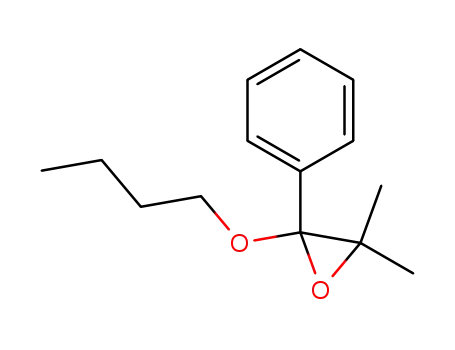
2-butoxy-3,3-dimethyl-2-phenyl-oxirane
-
100611-90-3
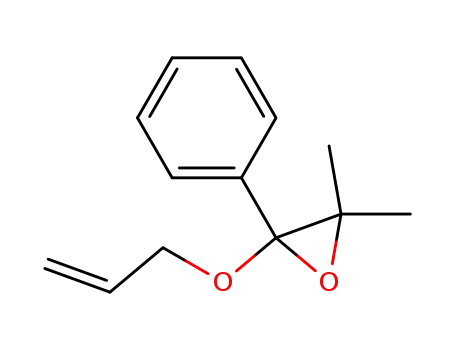
2-allyloxy-3,3-dimethyl-2-phenyl-oxirane
-
13694-96-7
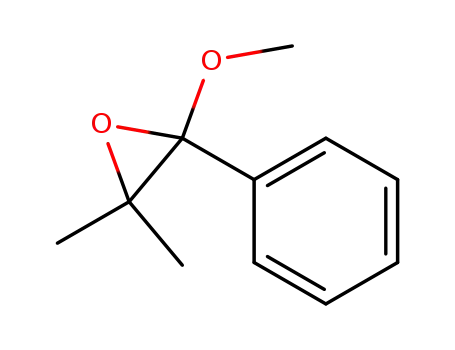
2-methoxy-3,3-dimethyl-2-phenyl-oxirane
Relevant Products
-
3-BROMO 2-HEXANONE
CAS:29584-99-4
-
Trimethylamine hydrochloride
CAS:593-81-7
-
2-Phenylpropan-1-amine hydrochloride
CAS:20388-87-8